Valved Spacer Manufacturers And PCD Company
Valved Spacer manufacturers and PCD Company – Valved Holding Chambers (VHCs) are medical devices approved by the Food and Drug Administration and available by prescription. Spacer/VHC helps to improve inhaled medication delivery to the lower airways. This is achieved primarily by enhancing hand-mouth coordination with the use of metered-dose inhalers (MDI), which reduces oral deposition of the particles and increases the fraction of droplets. Going for such products requires the best company and in this aspect, Luckys Pharma is the best Valved Spacer manufacturer and PCD Company.
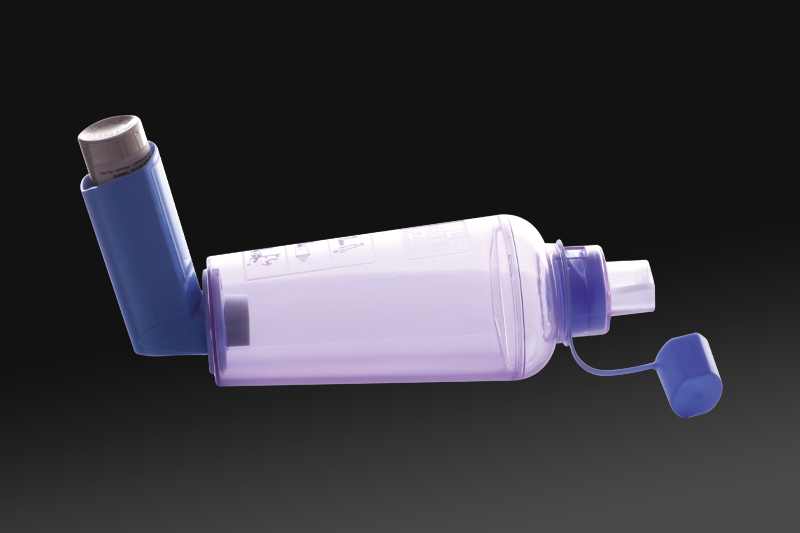
On the mouthpiece of an MDI, the spacer is a generic term for any open tube placed to extend its distance from the mouth. Often, a VHC has a whistle to provide feedback on correct inspiratory flow.VHCs are preferable to spacers due to their one-way valve and whistle.
Valved Spacer manufacturers and PCD Company | Luckys Pharma
Luckys Pharma is one of the top-notch Valved Spacer manufacturers and PCD Company that manufactures and delivers a massive range of products with utmost hygiene and safety aspects. We manufacture the best range of Valved Spacer segment.
The speed of inspiration is important in the Valved Spacer. If inhaled medication, such as corticosteroids, is inhaled too slowly, the medicine can remain in the mouth, leading to an increased risk of candidiasis. If the medication is inhaled too quickly, the medicine can be deposited in the back of the throat, leading to an increased risk for dysphonia (hoarseness).
Spacers/VHCs are made of either polycarbonate plastics or metal. Typically, they are shaped like a tube with openings at both ends; one opening is the mouthpiece that is placed into the mouth and the other opening is where the MDI is inserted.
Correct Use of the MDI with Spacer/VHC
- To make sure that there are no foreign objects that might be inhaled, inspect the mouthpiece.
- After removing the cap, shake the MDI and insert the MDI into the non-mouthpiece opening.
- After the start of a slow, deep breath, activate the inhaler ONE time immediately and continue to inhale for 3-5 seconds. Reduce the rate of inspiratory flow, if you hear the whistle until the whistle disappears; this is the correct inspiratory flow.
- Repeat as prescribed.
If a facemask is being used with a child or adult who cannot actively participate in medication-taking, hold the VHC with the facemask in place for 6 full breaths. This equals one puff. Repeat as prescribed.
Care and Cleaning
Manufacturers’ recommendations for care and cleaning will differ by the frequency of use, the type of device, and whether the spacer/VHC is made of plastic, non-static plastic, or metal. The directions below are general guides. To determine the correct care for a specific device, please refer to the manufacturer’s instructions.
Spacers/VHCs NOT Made of Anti-Static Plastics or Metals:
Disassemble all removable parts.
Wash by hand for 15 minutes and soak in diluted household detergent.
Do NOT rinse. Do not use bottle washers.
Allow drying thoroughly in a vertical position.
Reassemble and use as directed.
Spacers/VHCs Made of Anti-Static Plastic or Metals:
Disassemble all removable parts.
Wash by hand and soak in diluted household detergent for 15 minutes. Agitate gently.
Allow drying thoroughly in a vertical position.
Reassemble and use as directed.
Why are valved spacers important in respiratory treatment?
Valved spacers are essential devices used with inhalers to improve drug delivery efficiency and the key benefits are:
- Improve lungs
- Reduces drug wastage in the mouth or throat
- Ideal for children and elderly patients
- Helps in better coordination during inhalation
Who should use Valved Spacers?
- Asthma Patients
- COPD patients
- Pediatric patients
- Geriatric population
- Hospitals and clinics
- Homecare users
Why invest in Valved Spacer PCD Franchise?
The demand for respiratory devices is increasing due to over 262 million asthma patients globally and other reasons are:
- Growing chronic respiratory disease market
- Long term therapy
- Rising awareness of inhalation therapy
- Expanding home healthcare segment
What is the market size and growth of Valved Spacer?
The demand for valved spacers is increasing due to the rising burden of respiratory diseases like asthma and COPD. Here are the reports that explain the market of valved spacers:
|
The global inhaler spacer market from Wise Guy Reports, |
The market is valued at USD 2.1 billion in 2024 and projected to reach USD 3.5 billion by 2035 with a growing CAGR of 4.6%. |
|
The market is expected to grow from USD 2.4 billion in 2024 to USD 4.5 billion by 2034 with a growing CAGR of 5.9%. |
|
|
Another report from Research and Markets, |
The Global asthma spacers market grew from USD 2.07 billion in 2025 to USD 2.18 billion in 2026 and now it is projected to reach 3.03 billion by 2032 with a growing CAGR of 5.6%. |
What makes Luckys Pharma the Top-Ranked Valved Spacer manufacturer and PCD Company?
Luckys Pharma is a leading PCD franchise company. We manufacture Valved Spacers with the best quality of material, which ensures complete safety. Here we have listed below some of the amazing aspects:
- Luckys Pharma has an ISO 9001:2015 certification.
- Luckys Pharma conveys an outstanding scope of drug improvements, spanning across different medical areas.
- Quality is a significant need for Luckys Pharma. To guarantee patients, medical care establishments, and vendors get incredible quality items, it carries out inventive thoughts and assembly strategies.
- Throughout the excursion, the PCD establishment organization has achieved a number of achievements and recognition.
- To help patients get agreeable outcomes, all pharma items fulfill global quality guidelines. They offer customized pharma arrangements.
- In addition to business relations, the organization likewise offers syndication rights.
- Quality is the most extreme viewpoint for Luckys Pharma.
- For bundling to upgrade the time span of usability of items, Luckys Pharma executes demonstrated procedures.
Contact Details
Company Name: Luckys Pharma
Address: Plot No. 82, 1st Floor, Jaora Compound, Near Police Lines, Indore, M.P. India
Phone: 08889909734, 08889909733
Email: info@luckyspharma.com
FAQs
Who should use a valved spacer?
It is recommended for children, elderly patients and individuals who have difficulty using inhalers properly.
Are valved spacers reusable?
Yes, there are most valved spacers are reusable and can be cleaned regularly as per the manufacturer’s instructions.
What is the demand for valved spacers?
They are high in demand due to the rising number of respiratory diseases like asthma and COPD, and increasing awareness about effective inhalation therapy.
What documents are necessary to start a PCD pharma franchise with valved spacers?
If you have a valid drug license and GST registration, you can start a PCD pharma franchise.
Are valved spacers a profitable business for distributors?
Yes, it offers steady demand, which is essential for long term respiratory care and provides good margins, making it profitable for distributors.


